CARBON MONOXIDE IN THE REAL WORLD
Carbon monoxide is an important compound in the industry and in the home. However, tt can also pose serious environmental threats. Read more about it below!
How Carbon monoxide is used in industries

Carbon monoxide has many uses in multiple industries. For example, carbon monoxide is used as a semi-conductor in electronic chips. It is also used as a reducing agent for metals, because of it's high oxidization level; it is used to convert a naturally occurring oxide of a metal into a pure metal. An example of this would be when carbon monoxide is passed over hot iron oxide (pictured on the left), which creates metallic iron, converting it back to it's elemental form.
How Carbon monoxide is found in the home

Carbon monoxide is not used as a reactant in household appliances, as it is very dangerous. However, it is a naturally occurring by-product of combustion of fossil-fuels, so it's created by many household products, such as furnaces, stove-tops, cars, ovens, water-heaters, fireplaces, and clothes dryers. The amount of carbon monoxide deemed safe by the Occupational Safety and Health Administration is exceeded by the majority of household appliances that are fully functional, and that do produce carbon monoxide. These household products can cause home-dwellers to be poisoned by carbon monoxide if they break or become faulty. This is why the use of carbon monoxide is kept limited in homes, and that safety precautions are taken to avoid it.
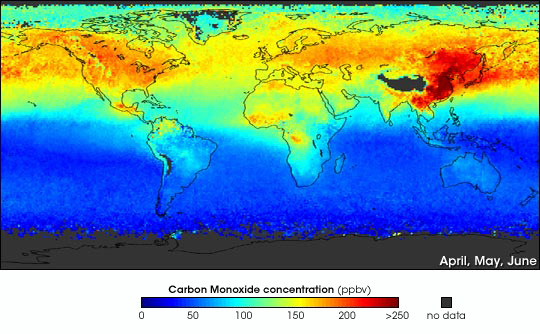
Effects of Carbon monoxide on the environnement
When carbon monoxide is created, it is normally disposed into the air that we breathe, contributing to climate change and the thickening of the ozone layer. Carbon monoxide is an unusual greenhouse gas as it stays around the area that it is produced from and doesn't circulate around the globe, unlike like carbon dioxide (CO2). Carbon monoxide itself doesn't have many direct impacts on the environnement as it is a weak greenhouse gas, but it has more indirect indirect impacts; carbon monoxide often effects the amount of other greenhouse gases in a region, such as methane, tropospheric ozone and carbon dioxide. Carbon monoxide also reacts with hydroxyl radical, (another weak greenhouse gas) to create carbon dioxide.